Launch excellence in Pharma needs both inspirational, innovative brand development, as well as agile launch deployment, and they clearly need to work hand in hand: this article focusses on how to get the most out of your Brand Development Capability.
We believe that brands of the future face a higher bar; stakeholders demand not only a better clinical profile, but also added value services and solutions that are affordable and ease the burden of long term disease management.
We are aware of the increasing opportunities to support the patient and their care teams with information, technology and interventions before, after and during treatment, and the resulting increasingly sophisticated solutions being launched.
And often these solutions are developed in partnership with third parties, beyond the PharmaCo.
As a result, best practice brand development breaks from the past in two areas, and teams need to think more broadly in two areas:
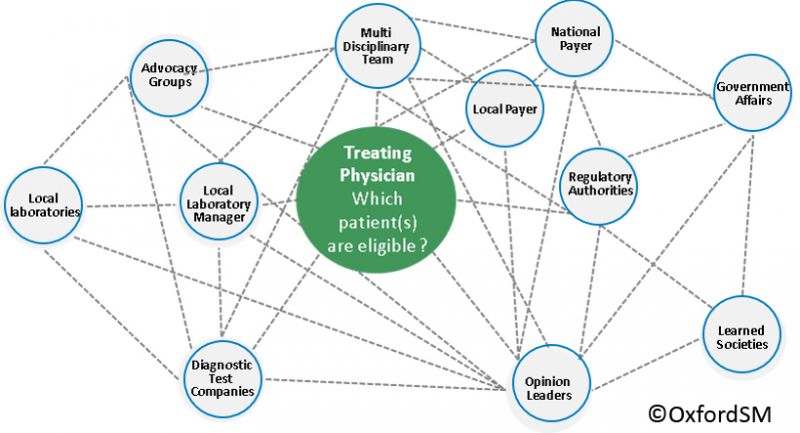
- Going beyond clinical trial data to deliver the value proposition – use Real World Evidence, Patient Advocacy and Direct Patient Feedback, in addition to trial data to shape the Value Proposition early in the development process
So, in the new world, the Value Proposition has to deliver against a range of needs beyond those of the regulatory bodies and these needs must be understood and fed in to the evidence plan before Phase 3 trials are locked down. This means that cross-functional teams need to create value propositions by customer type and then identify how they will deliver the evidence for each as part of their holistic Phase 3 planning

So Brand Development leaders have new opportunities, and their teams new challenges, in leveraging these opportunities.
Focussing on these in 5 areas in Brand Development, will help them do this:
- Optimising the product profile: early involvement to ensure TPP meets all stakeholder needs.
- Ensuring access: innovative pricing and market access models, supported by data collected via open access trials and electronic health records
- Patient identification: early development of diagnostic and prognostic tools and use of big data
- Building positive patient experiences across the journey: and in particular, establishing value with high responders first, especially in new markets
- Remain competitive long term: start early with the LCM, challenging how to enhance the value proposition.
Think of the opportunities for earlier detection, risk stratification, and treatment optimisation by the application of AI to big data, driving precision medicine.
This will, of course, require new skills such as open collaboration, agile ways of working, and effective partnering. Those organisations that meet this challenge, will increase their chances of faster clearance, accelerated adoption, and longer life for their franchise brands.
Next we’ll complete our review of how to set up in Pharma for a perfect launch, by considering Launch Deployment.
Meet us
If you’d like to talk more about how we can help develop your launch capability, please do get in touch. We’d love to hear from you - sarah.langan@oxfordsm.com
No Comments Yet
Let us know what you think